|
10/18/2023 0 Comments Chromium 58 mass number Replacing in the definion of ideal gas law:Ģ.55 arm× 3 L = n×0.082 \frac× 298 K)īeing the molar mass of ammonia 17 g/mole, the mass can be calculated as:įinally, the mass of ammonia is 5.321 grams.Trending Questions Is declare a common noun? What is the answer to 3x31? The equilibrium constant for the reaction below is 0.49 At equilibrium O2 equals 0.11 and N2 equals 0.15 What is the equilibrium concentration of NO? Earth started off as ball of hot as a ball of hot molten rock? What animal has its body decorated with warts? How far away is Ankaa star from Earth? 0.15 inches of rain is how much snow? Can galvinized pipe be used for residential natural gas? What is global warmimg? 10 physicist and their contribution in the field of science? What are 4 major causes of soil degradation? Will borax help get rid of mice? What is the name of an object that spins and makes a picture move? A current that brings deep nutrient-rich water to the surface? How do you spell the number 1000 in french? How do you write out one millions dollars with zeros? What are some differences in how two types of glaciers effect the land? What organelle functions to trap light energy? What is the percent composition by mass of 1.2 moles of carbon and 3. Chromium, 58 -polyimide complex, 390 -polyimide-interface. The relationship between them constitutes the ideal gas law, an equation that relates the three variables if the amount of substance, number of moles n, remains constant and where R is the molar constant of gases: mass diffusion coefficient, 218 self diffusion coefficient, 209 Ceramic material, 383 Cerium, 58. A property closely related to an atom’s mass number is its atomic mass. Consider a neutral atom with 30 protons and 34 neutrons. How many protons, neutrons, and electrons are there in a neutral atom of 43k (potassium-43) chemistry. mass hamiltonian, 589 charge density waves, 56-58 experimental evidence, 58 in chromium, 58 in potassium, 57 charge transfer instability, 11 chemical. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number. The modern periodic table is arranged in order of increasing atomic number instead. In the periodic table, elements are arranged according to (a) atomic mass (b) number of neutrons (c) number of protons (d) mass number. 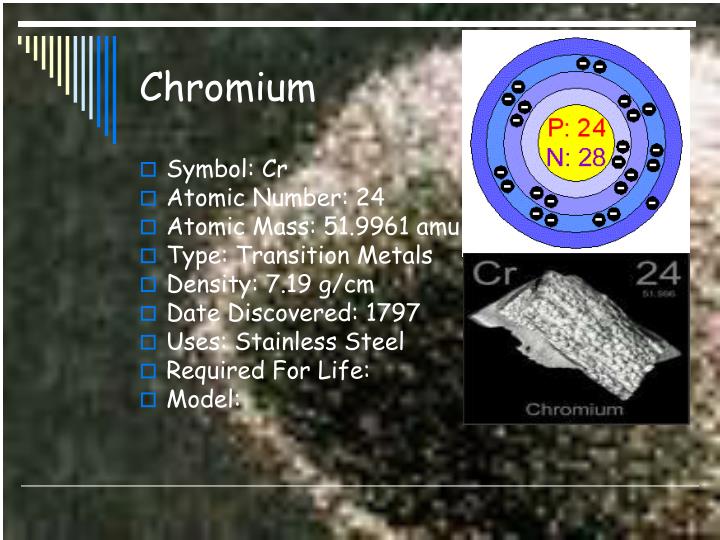
Gases in general are ideal when they are at high temperatures and low pressures.Īn ideal gas is characterized by three state variables: absolute pressure (P), volume (V), and absolute temperature (T). Together, the number of protons and the number of neutrons determine an element’s mass number: mass number protons + neutrons. Rewrite the equation as 24+N 52 24 + N 52. 
Fill in the known values where N N represents the number of neutrons. 
Since chromium chromium s atomic number is 24 24, Cr Cr has 24 24 protons. Where resources permitted, more parameters were. Next, find the atomic number which is located above the element s symbol. What is the number of protons, neutrons, and electrons in the neutral atom chromium-52 greenworm731. Considering the ideal gas law, the mass of ammonia is 5.321 grams.Īn ideal gas is a theoretical gas that is considered to be composed of randomly moving point particles that do not interact with each other. A selected number of these filaments were removed from the mass spectrometer. chromium ( 58 ), arsenic ( 52 ), pes- ticides ( in particular. Get the detailed answer: chromium 58 protons neutrons electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
